Nanocharacterization laboratory

Nanocharacterization Laboratory
The laboratory consists of several groups concentrating on Quantum sensing, Ultrafast time-resolved spectroscopy of photosynthesis, Spectroscopy of quantum dots and Nanocharacterization laboratory.
Our laboratory primarily concentrates on interactions between nanoparticles and nanostructured materials with the biological environment. Our primary tool of exploration is Atomic force microscopy combined with micro-spectroscopy. Due to the nature of our research, we are working on the intersection between physics, chemistry, and biology, leading to a broader research topic portfolio.
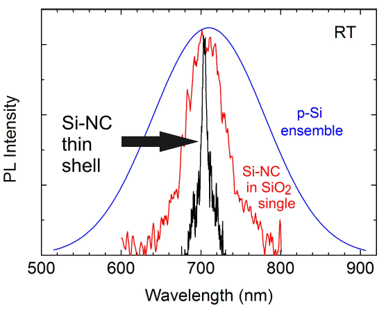
Synthesis, characterization, and study of protein corona formation on nanoparticles — from ensemble methods to the single-particle level.

AFM-based study of mammalian and plant cells — topography, surface adhesion, and mechanical properties.
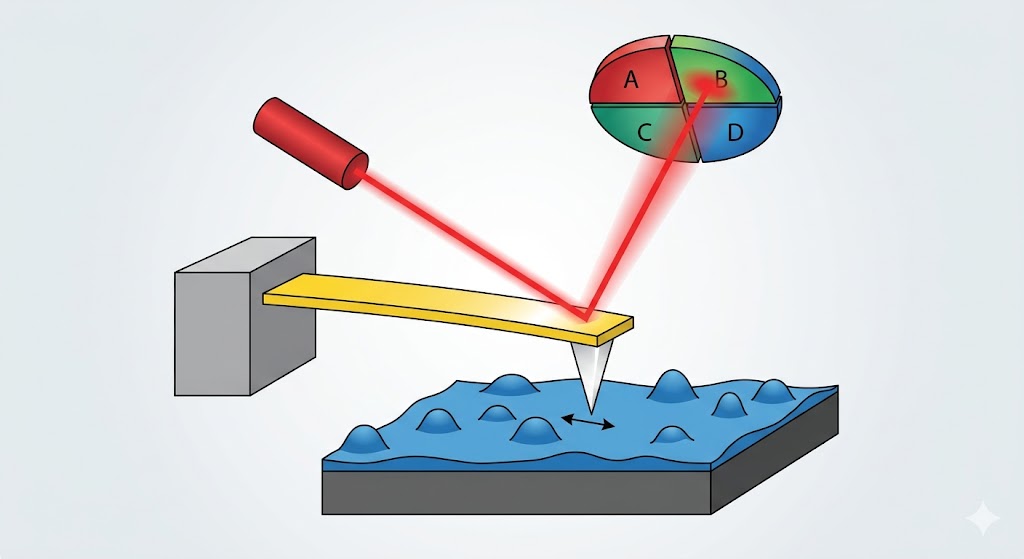
Large-area surface scans, nano-drug formulation characterization, and advising on nanoparticle synthesis and nano-interactions.
Research topics
Nanoparticles. Our laboratory holds or has taken part in several patents on nanoparticle synthesis and modifications. We are capable of nanoparticle synthesis and their chemical and physical characterization.
Protein corona development on nanoparticles. The protein corona determines the fate of nanoparticles in living organisms. It is still studied using outdated, ensemble-based methods that omit nuances in protein corona formation. In project GAČR 24-11110S (2024–2026), we concentrate on studying both single-nanoparticle protein corona formation and ensemble studies in order to reveal the little nuances by which protein coronas are formed.
Mammalian cell lines mechanoscopy. Our AFM is capable of measuring standard force curves, but also peak force curves, allowing detection of subtle changes in cell health and membrane behavior.
Plant cell studies. Our group is part of the core team of COST project CA24116 (2025–2029), where AFM complements the information between TEM/SEM and optical microscopy. We provide complex data not only regarding sample topography but also its adhesiveness and surface mechanical properties.
Contract research. Nanocharacterization of various surfaces on a large scale (zones 100 µm × 100 µm with resolution down to 0.1 nm in the Z axis), including mechanical properties. Characterization of nano-drug forms including physicochemical characterization (size, charge, stability). Advising and problem-solving with nanoparticle synthesis, nano-interactions, and nano-chemistry and physics at the nanoscale. Prices depend on the complexity of the given task.

The instrument
Our core instrument is an AFM NanoWizard 3 (JPK) with a PeakForce upgrade (Bruker), mounted above an inverted optical microscope (Olympus IX-73). The top port supports an EM-CCD camera (ProEM1024); the side port is coupled to a micro-spectroscopy setup with a monochromator (Acton SpectraPro-2358i) and a nitrogen-cooled CCD camera (Princeton Instruments Spec-10:400 DDBI).
Optical excitation is available at 405 nm (pulsed, <2 ns rise/fall), 633 nm, 473 nm, and 375 nm (CW), as well as white light and various LED sources, all coupled into the same optical path.
- Large-area scans: up to 400 µm × 400 µm
- Vertical resolution: down to 0.1 nm
- Modes: topography, adhesion, mechanical properties

Available devices
The laboratory is equipped for chemical and biological sample preparation.
- PCR cabinet
- Cell incubator
- Standard chemistry bench with fume hood
- DI water source
- Precision scales
- Absorption spectrometer Specord 250 (Analytic Jena)
- Oven up to 2500 °C with inert atmosphere
- Hot plate up to 500 °C
- Tube furnace (Carbolite Gero) up to 3000 °C with inert atmosphere


